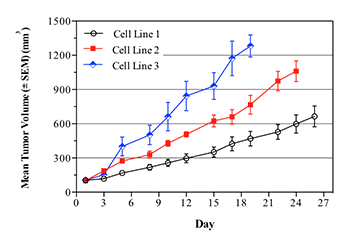
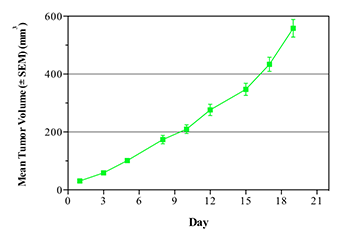
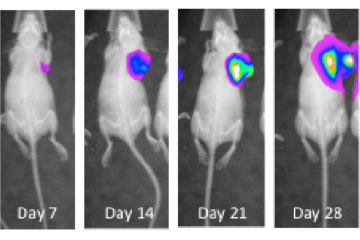
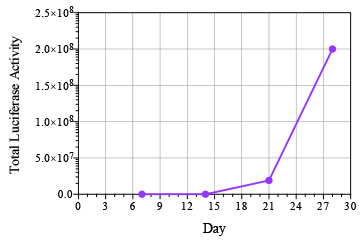
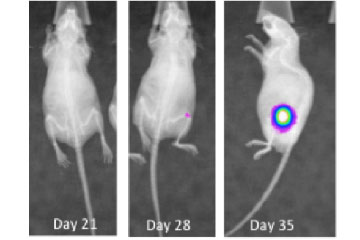
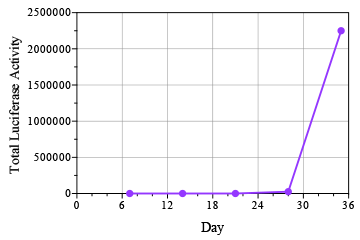
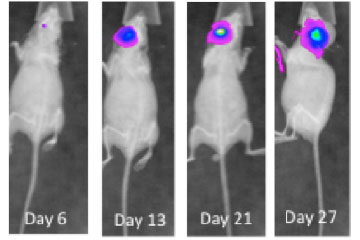
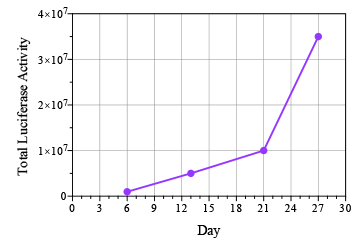
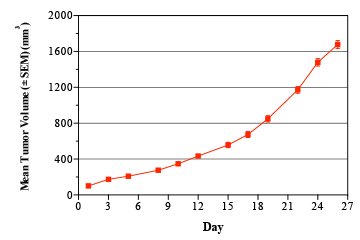
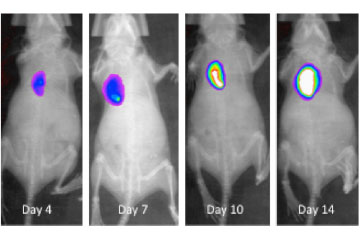
Breast cancer remains the leading cause of cancer in women, accounting for almost a quarter of cancers in women and more than one in ten cancer deaths in women.
Annually, there are an estimated 1.7 million new diagnoses of breast cancer worldwide, and despite recent advances, breast cancer remains a very significant clinical problem. According to the World Health Organization (WHO), over 508,000 women died in 2011 because of breast cancer.
Breast cancer is a form of cancer that originates in the epithelial tissues of the breast. Breast cancer accounts for 22.9% of all cancers in women, causing 458,000 deaths worldwide in 2008 (13.7% of cancer deaths in women).1-3 Most cases of breast cancer occur in the developed world, with an estimated 230,000 cases diagnosed in the United States annually and a similar number in Europe.
Improvements in diagnosis and treatment have raised 5-year survival rates in the developed world to between 80% and 90% in recent years, but survival rates in the developing world remain much lower. The primary risk factor for breast cancer in women is older age – the incidence of breast cancer increases with age. Additional risk factors include higher hormone levels and obesity. There are a number of chemical exposures including diet and smoking that have been linked to breast cancer, and a number of genetic defects have been linked to a higher incidence of breast cancer, most notably BRCA-1 and BRCA-2.4 However, the number of cases of breast cancer that can be directly attributed to genetic factors is low, and it is thought that environmental factors are the major contributor to breast cancer etiology after age and gender.
Traditionally, breast cancer has been graded based on histological characteristics such as degree of tissue organization, the uniformity of the nuclei, with grade increasing as the cells lose the typical characteristics of breast epithelium. More recently, treatment and prognosis have been related to receptor status, where the presence of estrogen receptor (ER), progesterone receptor (PR) and the human epidermal growth factor receptor 2 (also known as Her2, Neu, Erb-2 or CD-40).
Tumors that express the estrogen receptor can be treated with drugs that block this receptor, like tamoxifen, and typically carry a better prognosis.5 Tumors that are positive for Her2/neu generally have a worse prognosis than ER+ tumors and can be treated with antibodies that target Her2/Neu (Herceptin, Pertuzumab)6 and vaccine based approaches with compounds like Neuvax. In addition, these tumors can be treated with drugs that target the Her2/neu signaling pathway through PI3K/akt. Progesterone receptors expression is correlated with positive responses to ER-based therapy, but is not a therapeutic target.7 Tumors that do not express any of these three receptors are referred to as triple negative breast cancer, and represent a diverse group of tumors, including types that are found predominantly in younger women, are typically more aggressive, and consequently have a poor prognosis. However, the triple negative category also includes several rarer forms of breast cancer that do not share this poor prognosis.
In addition to receptor targeted therapies, most breast cancer patients receive some form of radiation therapy to the local area after surgery, followed by a course of conventional chemotherapy with cytotoxic agents, such as cyclophosphamide, Adriamycin (doxorubicin), cisplatin, 5-FU or a taxane (docetaxel or paclitaxel).