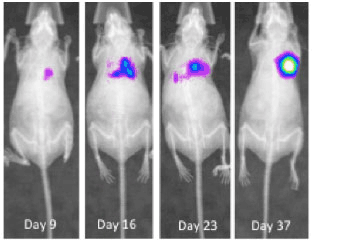
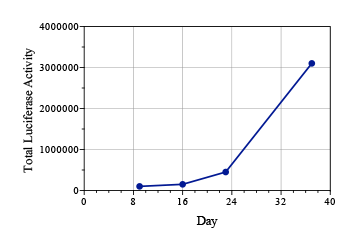
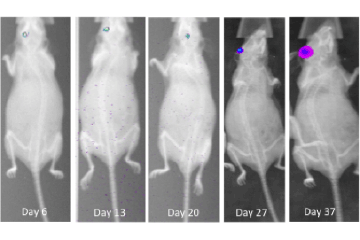
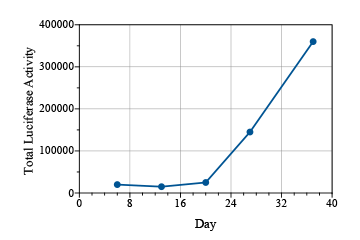
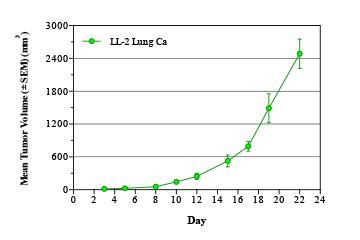
Lung cancer is the most common cancer worldwide, with an estimated 1.8 million cases annually.
Although lung cancer is largely attributable to smoking cigarettes, or secondary exposure to tobacco smoke, the number of cases of lung cancer remains high in the developed world and is rising rapidly in the developing economies as affluence makes tobacco products more affordable. As a result, lung cancer is likely to remain a significant cause of death worldwide for the foreseeable future. Five-year survival for patients with lung cancer is 15%, meaning that there is ample opportunity for new treatments to improve the overall prognosis of lung cancer patients.
Approximately 208,000 people are diagnosed with lung cancer in the United States annually and approximately 158,000 people die from lung cancer each year.
Between 80 to 90% of lung cancer cases are caused by smoking tobacco. Lung cancer accounts for 14% of all cases of cancer in the US annually.1 Overall 5-year survival for patients with lung cancer is 15%.2 Diagnosis is generally based on chest X-ray and treatment, while generally determined by the type of lung cancer, usually involves both radiation therapy and chemotherapy.
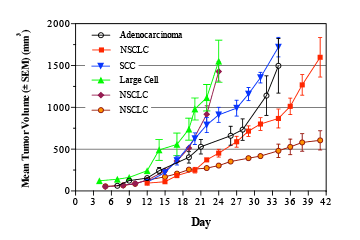
Lung cancer is broadly classified into two main types based on the cancer’s appearance under a microscope: non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC). Non-small cell lung cancer accounts for 80% of lung cancers, while small cell lung cancer accounts for the remaining 20%.3
NSCLC is further divided into four different types:
- Squamous cell carcinoma. (SCC) also known as epidermoid carcinoma. The most common type of NSCLC squamous cell carcinoma forms in the lining of the bronchial tubes.
- Adenocarcinoma. Adenocarcinoma is the most common type of lung cancer in women and in nonsmokers, adenocarcinoma forms in the mucus-producing glands of the lungs.
- Bronchio-alveolar carcinoma. This type of lung cancer is a relatively rare adenocarcinoma that forms near the lungs’ air sacs.
- Large-cell undifferentiated carcinoma.This rapidly growing cancer is composed of large cells forming an undifferentiated carcinoma typically near the outer edges or surfaces of the lungs.
Treatment for NSCLC typically involves surgery4 often with adjuvant chemotherapy usually platinum based (cisplatin, carboplatin), or taxanes (docetaxel, paclitaxel) and cytotoxic agents (gemcitabine, etoposide).5-7
Treatment for SCLC is generally by radiation and/or chemotherapy and does not commonly involve surgery.3 Chemotherapy usually consists of cisplatin and/or etoposide and combinations of these two compounds with paclitaxel, gemcitabine, vinorelbine and topotecan or irinotecan are also commonly used.8-10
Numerous alterations to oncogene expression have been reported in lung cancers.
As in many malignant tumors, lung cancers show a high degree of genetic instability. Activating mutations in the KRAS gene are seen in 25-40% of lung cancers . Approximately 3% of lung cancer patients have activating mutations in Braf, while 2% have activating mutations in the MEK/MAPK-1 gene, which lies in the same pathway. Both Braf and MEK lie in the Ras oncogene pathway, downstream of ras.
Reports indicate that 10-15% of lung cancers have activating mutations of the epidermal growth factor receptor (EGFR),11 while 2-4% of cases have activating mutations of the related gene HER2. Activations of the Ras and EGFR/Her-2 are almost invariably mutually exclusive, suggesting that each serve as driver mutations for the cancer, rather than incidental mutations that have occurred during the development of the tumor. Mutations in the Met and ALK oncogenes are found in relatively small percentages of patients (1%-7%) and are generally not found in in patients with activations of KRAS or EGFR/Her-2. Activation of the PI3K/AKT/mTOR pathway is seen in approximately 60% of NSCLC cases, but is generally seen in conjunction with either KRAS or EGFR/Her-2 activation and is thought not to be a primary driver of tumor growth. Additionally, mutations have been identified in the Ros-1, RET, FGFR1, and DDR2 oncogenes, as well as numerous tumor suppressor genes, but the exact incidence and role of these mutations in the development of lung cancer is still under investigation.