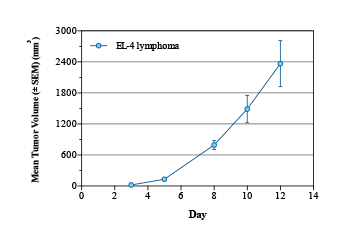
Lymphoma is a cancer of the blood cells like that of leukemia and is the seventh most common type of cancer.
Unlike leukemia, lymphoma consists of solid tumors derived from white blood cells known as lymphoid cells (or lymphocytes). Most lymphomas are currently classified as Hodgkin lymphoma (HL) or non-Hodgkin lymphoma (NHL). Examples of other lymphomas are multiple myeloma and immunoproliferative disease. There are an estimated 566,000 cases of lymphoma worldwide each year, with 300,000 people lymphoma deaths annually.
Although NHL accounts for approximately 90% of all lymphoma, it is divided into a wide variety of subtypes, including precursor T-cell lymphoma, follicular lymphoma, diffuse large B-cell lymphoma, mantle cell lymphoma, Burkitt’s lymphoma and MALT lymphoma. Lymphoma can occur almost anywhere in the body, but is most commonly associated with lymph nodes. The clinical picture is further complicated by the grade and site of the tumor, making treatment very patient specific.
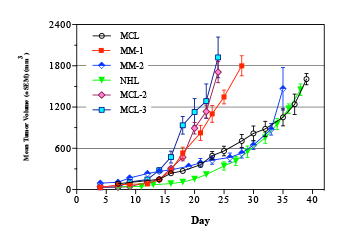
Mantle Cell Lymphoma (MCL)
Mantle Cell Lymphoma (MCL) is a B-lymphocyte malignancy that afflicts approximately 15,000 people in the US, but has a higher prevalence in Europe. Mantle Cell Lymphoma is one of the rarest of the non-Hodgkin’s lymphomas (NHLs), comprising about 6% of NHL cases.1 Mantle Cell Lymphomas are named for the mantle zone of lymph nodes that they typically infiltrate, and are characterized by a t(11;14)(q13;q32) chromosomal translocation2 which results in the overexpression of cyclin D1.3 Overexpression of cyclin D1 disrupts the normal cell proliferation cycle, and prevents programmed cell death (apoptosis) that would limit the proliferation of normal cells.4 Consistent with the putative origins of Mantle Cell Lymphomas in the immature B-cells of the mantle zone of lymph notes, Mantle Cell Lymphomas typically express the cell surface antigen CD5.
Mantle Cell Lymphoma is an aggressive variety of lymphoma, and is divided into two main cytological variants; typical or blastoid. Typical cases are small to intermediate sized cells with irregular nuclei. Blastoid variants have intermediate to large sized cells with finely dispersed chromatin and are more aggressive in nature. Historically, Mantle Cell Lymphomas have poorer prognoses than other lymphomas. However, recent improvements in chemotherapy have increased median survival from around 3 years to 5-7 years, largely due to the combination of newer drugs, like Rituxan, with conventional combinations of cytotoxic agents.5
Multiple Myeloma
Multiple myeloma is a B-cell malignancy, characterized by collections of abnormal plasma cells (mature B-lymphocytes) in the bone marrow, where they interfere with the production of normal blood cells. Most cases of multiple myeloma also result the production of an abnormal antibody (or paraprotein) that accumulates in the blood and can cause kidney problems. Bone lesions and high calcium levels are also frequently seen in multiple myeloma.6,7
There are approximately 22,000 new cases of multiple myeloma annually in the United States, and approximately 71,500 in the US living with the disease. Multiple myeloma is the second most common hematological malignancy (after Hodgkin’s lymphoma), representing 1% of all cancers, and 2% of all cancer deaths. Initial diagnosis peaks between the ages of 65 and 70, with more men than women affected. In the United States, African Americans have the highest incidence of multiple myeloma at 9.5 cases per 100,000 while Asian Americans have the lowest. The incidence of multiple myeloma among Americans of European heritage is 4.1 per 100,000. With current therapy, median survival is 3-4 years.
The diagnosis of multiple myeloma is based on the key characteristics of the disease; occupation of the bone marrow cavity – the presence of space occupying bone lesions and the production of paraprotein. The expansion of the multiple myeloma in the bone marrow causes a decrease of normal blood cell production, which results in anemia (decrease in red blood cells) and an increase in infections (decrease in white blood cells). These symptoms can be evaluated through blood cell counts and an evaluation of bone marrow function.
The production of paraprotein can cause renal failure due to the high levels of free light chains in the blood. These symptoms are evaluated through measurement of kidney function and the presence of high levels of paraprotein in plasma and urine. The bone lesions frequently cause bone pain, and lead to hypercalcemia due to loss of calcium from the bone. Bone pain is the most frequent symptom of multiple myeloma, typically due to the activation of osteoclasts by the tumor cells, resulting in excess resorption of bone leading to fractures and bone pain. In addition, the changes in the bone can lead to pressure on the nerves including the spinal cord, resulting in a variety of neurological symptoms depending on the nerves affected.
On top of these symptoms, the hypercalcemia can also cause weakness, confusion and fatigue due to its impact on CNS function. Diagnosis of these conditions is confirmed by serum calcium levels and bone X-rays. The staging of multiple myeloma is currently based on serum levels of β2-microglobulin (which increase with stage) and albumin (which decrease with stage).8
Multiple myeloma is a cancer of mature B-lymphocytes (as evidenced by the production of immunoglobulin), and is frequently characterized by a chromosomal translocation between the immunoglobulin heavy chain (chromosome 14 locus q32) and one of a number of oncogenes, resulting in the aberrant expression of the oncogene, and the expression of free light chain (which has no heavy chain to bind to). This translocation is thought to be the initiating event for the majority of multiple myelomas, resulting from an error during the immunoglobulin heavy chain V-D-J recombination that occurs when B-cells become activated.9
Treatment of multiple myeloma combines bisphosphonates (which treat the bone loss associated with the bone lesions) with anti-cancer drugs (vincristine, doxorubicin, melphalan) and immunosuppressive drugs (cyclophosphamide, thalidomide, dexamethasone and other steroids). Bone marrow or hematopoietic stem transplantation is also frequently used in the treatment of multiple myeloma.10