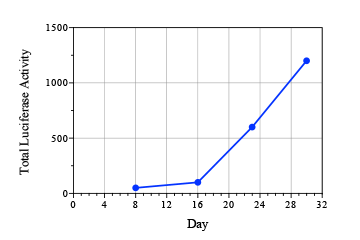
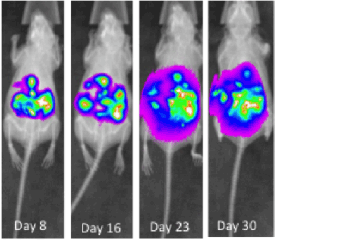
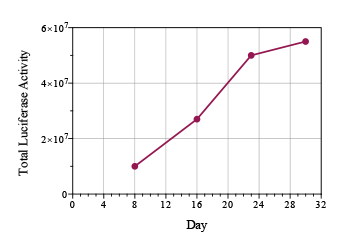
Prostate cancer is cancer that develops in the prostate gland of the male reproductive system.
Prostate cancer is diagnosed in 1.1 million men worldwide each year, and causes 307,000 deaths. Prostate cancer is the second most common cancer in American men (after skin cancer) with 233,000 cases diagnosed annually in the United States. It is the fifth leading cause of cancer death, causing 29,480 deaths annually. About 99% of prostate cancer occurs in men over the age of 50 and the vast majority of cases are seen in men over the age of 60.
There are an estimated 2.7 million men worldwide living with a diagnosis of prostate cancer, and 15% of men will be diagnosed with prostate cancer at some point during their lives.
Worldwide, there are 1.1 million cases of prostate cancer annually and 307,000 deaths. 99% of prostate cancer occurs in men over the age of 50 and the vast majority of cases are seen in men over the age of 60.1 The vast majority of cases of prostate cancer are asymptomatic, with studies of men over 60 who died from other causes revealing an incidence of undiagnosed prostate cancer of 30-70% depending on the study.2
The diagnosis of prostate cancer by either PSA blood test or rectal exam is controversial, since many cases of prostate cancer would normally remain asymptomatic during the patient’s lifetime, and the diagnosis of asymptomatic prostate cancer is thought to lead to over-treatment. The 5-year survival rate for prostate cancer is approximately 99%.3 Consequently, clinical management of patients with prostate cancer is often limited to watchful waiting. Treatment of prostate cancer is by surgery, followed in most cases by radiation therapy along with hormone therapy (anti-androgen) and chemotherapy if there is evidence that the cancer has spread beyond the prostate. A wide variety of anti-cancer agents are currently used to treat prostate cancer, but treatment usually begins with a taxane (docetaxel).4
Treatment options for prostate cancer are generally based on a histological score known as a Gleason grade, after the pathologist who developed it. In this scoring system, the tumor is given 2 grades based on 5-point scoring scale of the dominant and secondary histological pattern. The final Gleason score is the sum of the 2 grades. Scores higher than 7 are considered aggressive and treated accordingly, while scores of 5 or below are regarded as low grade and treated conservatively.5
Approximately 5% of prostate cancer is thought to have a hereditary basis, however the number of genes that have been shown to have increased expression in prostate cancer is high, suggesting that the etiology of prostate cancer is complex.6,7 These genes include cell cycle genes CDKN1B (cyclin-dependent kinase inhibitor 1β)8 and MCM7 (mini-chromosome maintenance component 7),9 cell surface receptors IL11Rα (Interleukin-11 receptor α) and LPAR6 (lysophosphaditic acid receptor 6),10 ion channels and transporters ABCB1 (ATP-binding cassette sub-family B member 1),11 KCNMA1 (potassium large conductance calcium-activated channel, subfamily M, alpha member 1), tyrosine kinases or MAP kinases, FGR (Gardner-Rasheed feline sarcoma viral (v-fgr) oncogene homolog), MAP3K7 (Mitogen-activated protein kinase kinase kinase 7),12 and Raf-1, cytoskeletal elements LCP1 (lymphocyte cytosolic protein 1) and VCL (vinculin). In addition, applifications are seen in N-Ras (8), transcription factors AR (androgen receptor), ERG (V-Ets Avian Erythroblastosis Virus E26 Oncogene Homolog),13 NCOA2 (nuclear receptor coactivator 2), NKX3-1 (Homeobox Protein NK-3 Homolog A) (8), and RUNX1T1 (runt-related transcription factor 1; translocated to, 1) as well as HACE1 (HECT Domain And Ankyrin Repeat Containing E3 Ubiquitin Protein Ligase 1), WWP1 (WW Domain Containing E3 Ubiquitin Protein Ligase 1),14 HMGN1 )High Mobility Group Nucleosome Binding Domain 1), PLAU (plasminogen activator, urokinase), and PSAP (prosaposin).
The overall picture of a genetically diverse cancer with an unclear relationship between biology and treatment options suggests a fertile field for target-based pharmaceutical development.